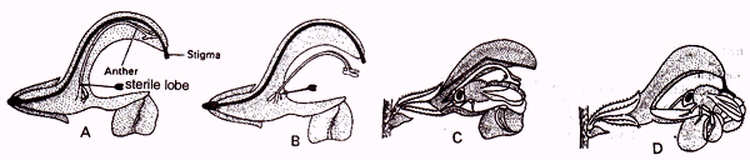ISC 12>CONTENT>STRUCTURE AND FUNCTION OF PLANTS>5. REPRODUCTION IN ANGIOSPERMS
SYLLABUS
- Vegetative reproduction: advantages and disadvantages of vegetative reproduction, micropropagation – plant tissue culture
- Sexual reproduction: development of male and female gametophytes, types of ovules, placentation,
- Pollination,: Advantages of self and cross-pollination
- Fertilisation (Amphimixis) and
- Formation of endosperm, embryo.
REPRODUCTION
- Reproduction is one of the most important features of living organisms.
- Reproduction is defined as a biological process in which an organism gives rise to young ones (offspring) similar to itself.
- The offspring grow, mature and in turn produce new offspring. Thus, there is a cycle of birth, growth and death. Reproduction enables the continuity of the species, generation after generation
- There are two basic forms of reproduction.
- Asexual reproduction
- Sexual reproduction
MODES OF REPRODUCTION IN ANGIOSPERMS
- In angiosperms or flowering plants, there are several modes of reproduction.
- Generally they are arranged in two groups.
- Asexual or vegetative :
- Sexual reproduction
A. VEGETATIVE PROPAGATION
- Vegetative reproduction is the process of multiplication in which a portion of the plant body becomes detached and develops into a new plant body.
- It is the simplest way of reproduction under favorable conditions.
- Year after after same variety can be produced.
- Since all the plants are genetically alike, they are susceptible to same diseases.
- Types : It is of two types.
- Natural vegetative propagation
- Artificial vegetative propagation
1. Natural vegetative propagation: It can takes place by the following methods.
A) Vegetative propagation by roots.
- The swollen tap roots of carrot, turnip and radish have buds at the base of old stems just above the tap root which serve as organ of vegetative propagation.
- Adventitious roots of Asparagus, Dahlia, sweet potato, yam etc. serve to propagate plant vegetatively
B) Vegetative propagation by stem
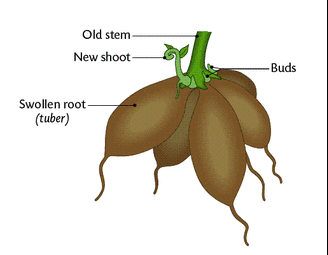
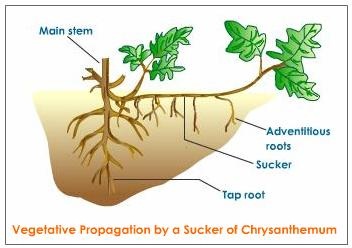
- (a) Subaerial stems: Subaerial stems may develop as lateral branches from the mother plant. This may break up from- the parent plant and then, grow into new plants. Example- Runners (Oxalis), sucker - (banana, Chrysanthemum), stolon (Jasmine), offset (Eichhornia)
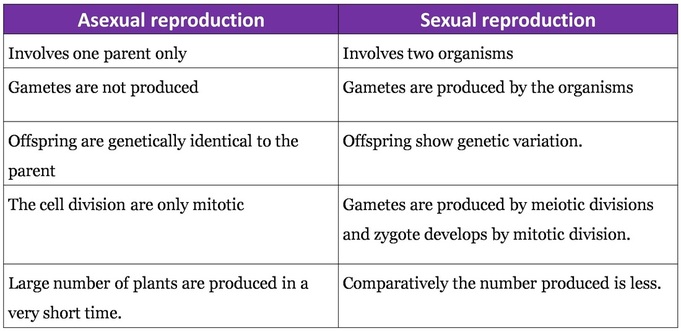
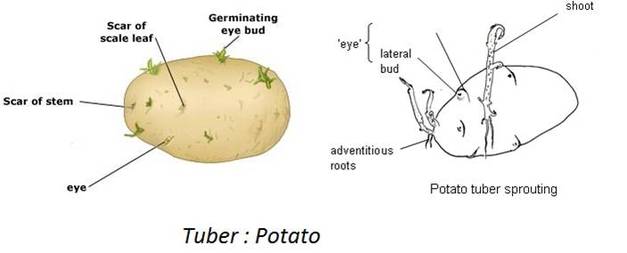
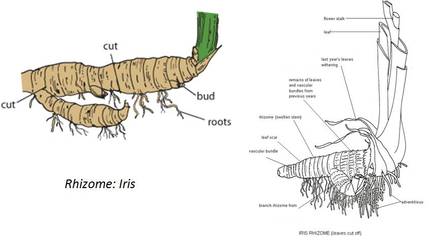
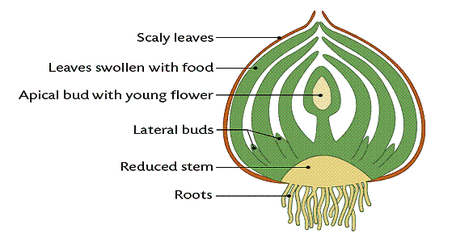
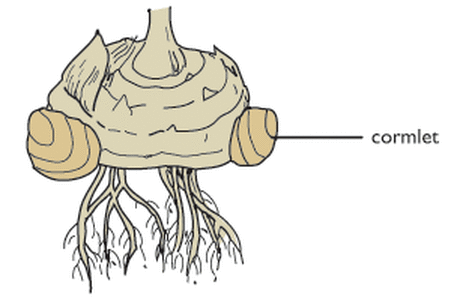
(b) Underground Stems: In certain plants the underground stems become modified for storage of food during the active phase of the growth. Examples- Rhizome (Ginger), tuber (Potato), bulb (Onion) and corm (colocasia)
a) Subaerial stem
|
Offset
|
b) Underground stem
|
Stem tuber: Potato
|
|
Bulb: Onion
|
|
Corm: Colocasia
|
C) Vegetative propagation by leaf
2. ARTIFICIAL VEGETATIVE PROPAGATION
Vegetative propagation by artificial means may be brought about by the following methods.
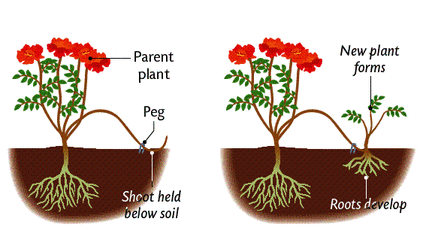
A. LAYERING
- In layering, the development of adventitious roots is induced on the stem before it is separated from the parent plant. In lemon, rose, jasmine, strawberry, raspberry, grape-vine, etc., the lower branch is bent down and covered under a light layer of moist soil by pushing the tip into the soft ground. After some time, adventitious roots develop and on cutting the branches from the parent plant, these develop into new plants. This is known as layering.
TYPES OF LAYERING:
|
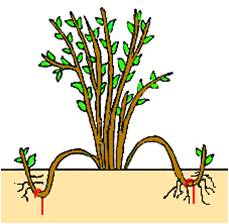
SIMPLE LAYERING
Simple layering means bending a branch to the ground and getting it to root where it touches This method is used mainly for shrubs with flexible branches,
such as Forsythia, Spirea, and Rambler Rose. |
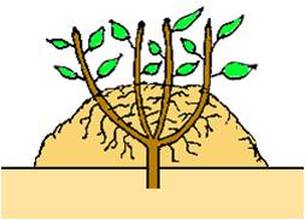
MOUND LAYERING
Mound layering is useful with heavy-stemmed, closely
branched shrubs. It is also useful for fruit root stock production. The original plant may be cut back to encourage many new shoots to grow from the base. Then, the following spring after the new shoots have grown approximately 8- 10 inches, mound soil containing sphagnum peat moss about 7-9 inches deep around the shrub.Roots will grow into the surrounding soil from the new growth. The following autumn or spring, gently dig into the mound, separate and transplant the new plants. |
AIR LAYERING OR GOOTEE
It is a modified fom of layering. In this case the cut or injured branch is not buried in the ground but is bound with mud and rags, etc., which is kept moist .The roots develop at this portion within a period of about a month or two. Now the branch is cut and separated from the parent plant from below the tied portion and is planted in the soil. This method is applied for the vegetative propagation of pomegranate, orange, lemon, guava, lokat, litchi, etc.
|
B. CUTTING
- In this process a vegetative part of a plant is taken and rooted to form a new plant.
- Several plants like sugarcane, rose, Duranta, Coleus and China-rose, etc., are grown from stem-cutting.
- A portion of stem with leaves is cut from the parent and placed in a suitable rooting medium for that particular species of plant. Some suitable media include moist sand, a mixture of peat moss and soil, or water used. After roots have developed from the cut end of the stem the cutting is transplanted to soil.
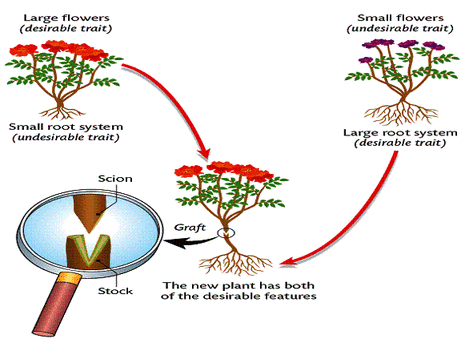
C. GRAFTING
(b) Whip grafting.
- In this case, a small branch of a plant is inserted into the stem of a rooted plant of the same or allied species. As a result of insertion, organic union or fusion of tissues takes place and both of them grow as one. The inserted plant is called the scion, the rooted plant the stock and the phenomenon is called grafting.
- The scion is a shoot, 4 -12 inches in length. Its all the buds are kept intact while all the buds of the stock are removed. The graft is placed on the stock and the joining portion is covered with a layer of wax or clay in order to prevent the evaporation of water and the entry of injurious bacteria. After some time the tissues of the scion and the stock become united. Grafting is of two types:
(b) Whip grafting.
- (a) In splice grafting, both graft and stock are cut across obliquely at about the same angle and then firmly tied together.
- (b) in whip grafting, both graft and stock are cut diagonally. Now a vertical notch is made in the stock and the graft is cut at one end to make a chisel-shaped structure or tongue. This tongue of the graft is inserted into the notch of the stock and the two are bound. In apple, citrus, mangoes, rose and lemon, new and superior plants are grown in this way.
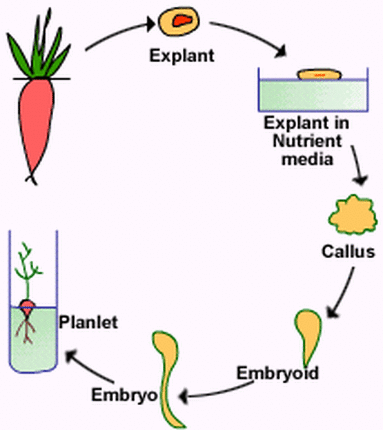
B. MICROPROPAGATION
(the production of a large number of individual plants from a small piece of plant tissue cultured in a nutrient medium)
Tissue culture:The aseptic culture of plant protoplasts, cells, tissues or organs under conditions which lead to cell multiplication or regeneration of organs or whole plants.
(the production of a large number of individual plants from a small piece of plant tissue cultured in a nutrient medium)
Tissue culture:The aseptic culture of plant protoplasts, cells, tissues or organs under conditions which lead to cell multiplication or regeneration of organs or whole plants.
|
Steps of micropropagation
|
ADVANTAGES OF PROPAGATION BY TISSUE CULTURE TECHNIQUE
- This technique enabled us to produce large number of plants in relatively short time.
- Disease free plants can be obtained by culturing the shoot apices (meristem) of infected plants.
- Very small size explants can be used for micropropagation. This is impossible with conventional technique. Important when limited explant is available.
- Material multiplied by micropropagation can be maintained in small place, packing and transport is also easy due to small size.
- Micropropagation is the only viable method of multiplying genetically modified cells or cells after protoplast fusion.
- In case of dioecious species, where one of the sex is more desirable then under such circumstances plants of desired sex can be selectively multiplied by this technique.
- The output is clean, healthy and pathogen free, as during micropropagation, fungi and bacteria are usually eliminated.
- Independent of the season; can be carried out through out the year.
LIMITATIONS :
- Requirement of sophisticated facilities
- High production cost
- Requirement of skill in handling and maintenance.
- Somaclonal variations may arise during in vitro culture when a callus phase is involved.
- For many valuable species suitable micropropagation techniques are not available (e.g. mango).
ADVANTAGES OF VEGETATIVE PROPAGATION
- The plants that cannot produce viable seeds such as banana, sugarcane, seedless grapes can be easily grown by vegetative propagation.
- It is easier,less expensive and a rapid method of propagation.
- Superior quality fruits or flowers can be produced by the method of grafting.
- Preservation of desirable characteristics due to no genetic recombination
- By tissue culture, a large number of of disease free identical plants can be grown in very short time.
DISADVANTAGES OF VEGETATIVE PROPAGATION
- Unwanted characters can not be eliminatedfrom plants.
- When plants grown repeatedly they may lose vigor
- May become susceptible to diseases.
- Vegetative parts such as root, stem leaves, bulbil etc can not be preserved for longer periods as they easily attacked by pathogens.
C. SEXUAL REPRODUCTION
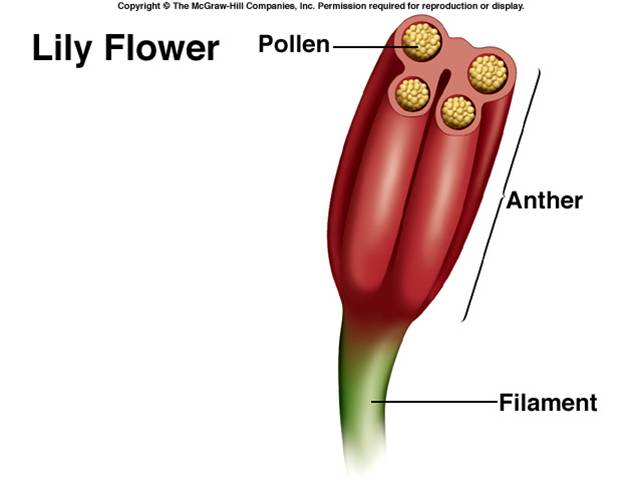
FLOWER AND FLORAL PARTS (Details in practical )
DEVELOPMENT OF THE MALE GAMETOPHYTE (POLLEN)
|
a) Structure of mature anther
|
b) Development of microsporangium
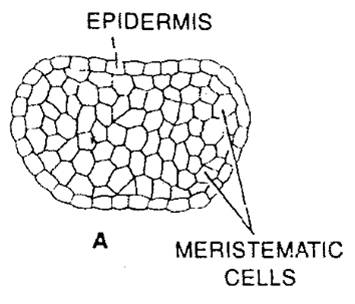
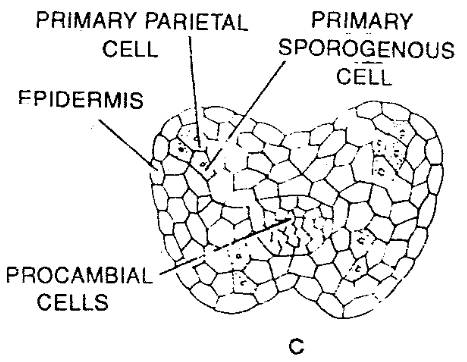
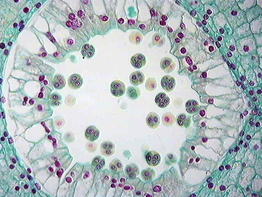
- Young anther appears to be a homogeneous mass of meristematic cells which is surrounded by epidermis. (Fig-a)
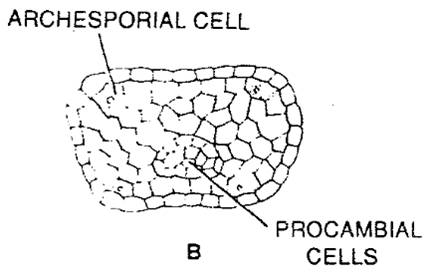
- It then becomes four-lobed and four rows of archesporial cells are differentiated. (Fig-b)
- The archesporial cells are marked off from the surrounding cells by their more deeply stained cytoplasm and conspicuous nuclei.
- Each of the archesporial cells now cuts off a primary parietal cell on the inner side. (Fig-c)
- The parietal cell now divides by periclinal and anticlinal walls giving rise to several layers of cells forming the wall of the anther.
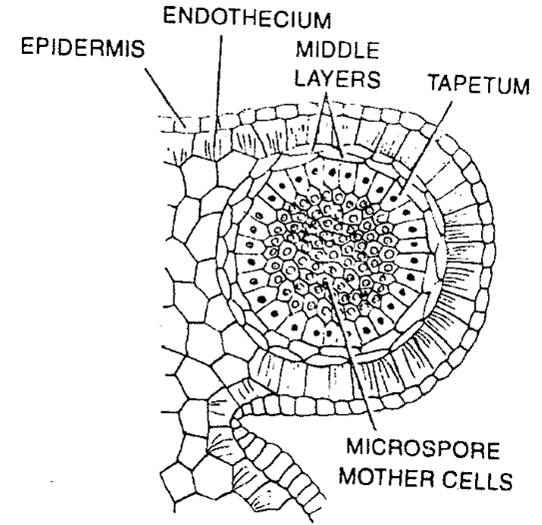
- Wall of anthers:
- Role of tapetum: It provides nourishment to the developing pollen grains. During microsporogenesis, the cells of tapetum produce various enzymes, hormones, amino acids, and other nutritious material required for the development of pollen grains. It also produces the exine layer of the pollen grains, which is composed of the sporopollenin.
- The sporogenous cell divides to give rise to a number of microspores or pollen mother cells.
Development of anther(Microsporangium)
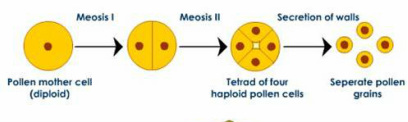
c) Microsporogenesis :
|
Microsporogenesis under microscope
d) Structure of pollen grains
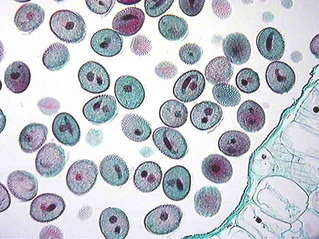
- The pollen grains represent the male gametophytes.
- There is variety of architecture – sizes, shapes, colours, designs – seen on the pollen grains from different species.
- Pollen grains are generally spherical measuring about 25-50 micrometers in diameter. It has a prominent two-layered wall.
- The hard outer layer called the exine is made up of sporopollenin which is one of the most resistant organic material known. It can withstand high temperatures and strong acids and alkali. No enzyme that degrades sporopollenin is so far known.
- Pollen grain exine has prominent apertures called germ pores where sporopollenin is absent.
- Pollen grains are wellpreserved as fossils because of the presence of sporopollenin.
- The inner wall of the pollen grain is called the intine. It is a thin and continuous layer made up of cellulose and pectin. The cytoplasm of pollen grain is surrounded by a plasma membrane
e) Development of male gametophyte
|
Figure: A
|
Figure:B
|
BQ: Explain the development of an anther and the formation of microspores in angiosperms. 4M (2006)
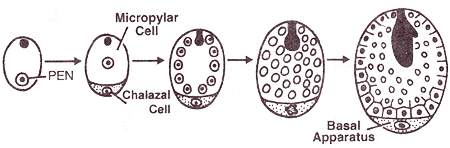
DEVELOPMENT OF FEMALE GAMETOPHYTE
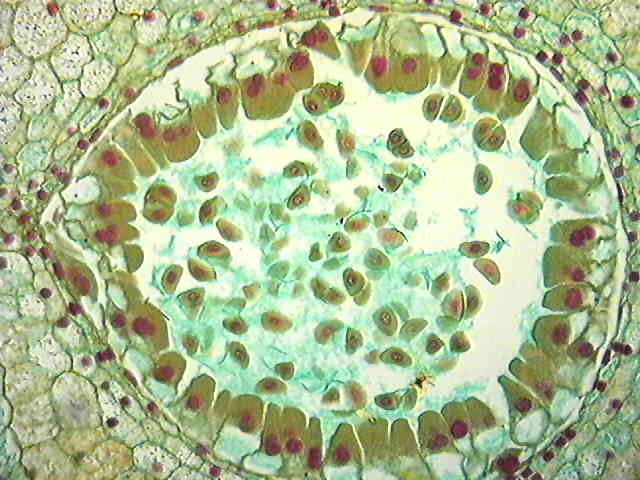
Structure of the ovule (Megasporangium)
|
Development of megasporangium
- The ovule develops as a small protuberance of the placental tissue.
- In every young ovule a single hypodermal cell is differentiated a the archesporial cell.
- The archesporial cell may divide to form a primary parietal cell and a primary sporogenous cell , or it may function directly as the megaspore mother cell.
- The primary parietal cell may remain undivided or it may undergo periclinal and anticlinal divisions to form a variable number of wall layers.
- The primary sporogenous cell usually functions as the megaspore mother cell without undergoing any further divisions.
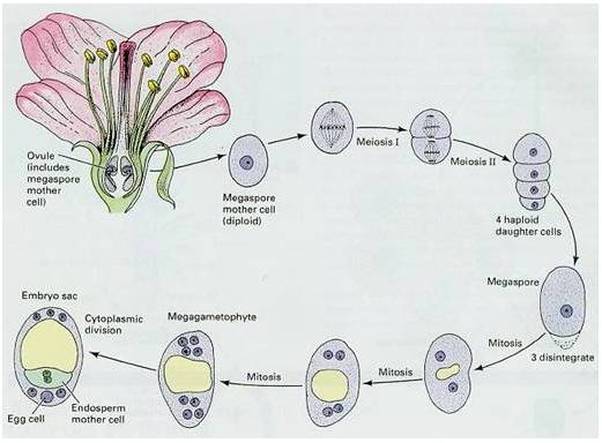
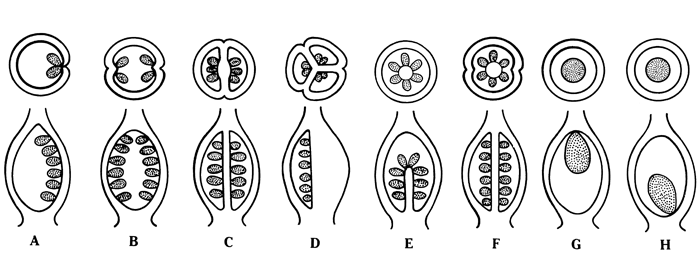
- Megaspore mother cell undergoes meiosis and a linear row of haploid megaspore cells is formed.
Megasporogenesis
- One of the nucellar cell in the micropylar region is differentiated into megaspore mother cell.
- The cell is larger, contains dense cytoplasm and a prominent nucleus.
- It undergoes meiosis forming 4 haploid cells called megaspore tetrad.
- 3 megaspores degenerate and only one megaspore become functional.
- Functional megaspore is the first cell of female gametophyte.
Megasporogenesis and development of female gametophyte
Development of female gametophyte
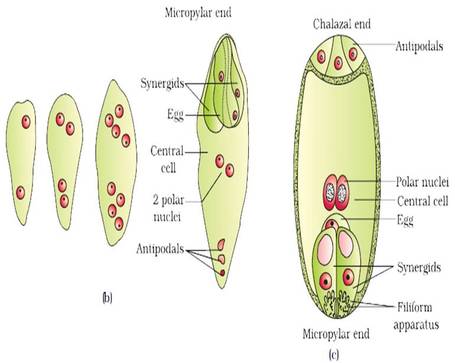
- The nucleus of the functional megaspore divides mitotically to form two nuclei which move to the opposite poles, forming the 2-nucleate embryo sac.
- Two more sequential mitotic nuclear divisions result in the formation of the 4-nucleate and later the 8-nucleate stages of the embryo sac.
- Nuclear divisions are not followed immediately by cell wall formation.
- After the 8-nucleate stage, cell walls are laid down leading to the organisation of the typical female gametophyte or embryo sac.
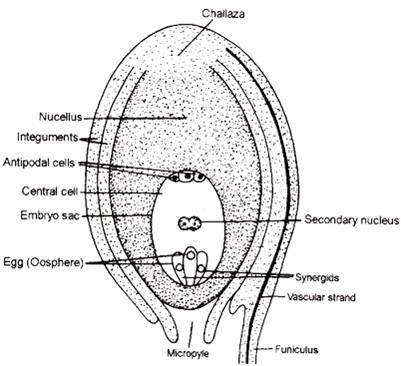
- Six of the eight nuclei are surrounded by cell walls and organised into cells; the remaining two nuclei, called polar nuclei are situated below the egg apparatus in the large central cell.
- Three cells are grouped together at the micropylar end and constitute the egg apparatus. The egg apparatus, in turn, consists of two synergids and one egg cell.
- Three cells are at the chalazal end and are called the antipodals.
- The large central cell has two polar nuclei. Thus, a typical angiosperm embryo sac, at maturity, though 8-nucleate is 7-celled.
BQ: Describe the development of female gametophyte in angiosperms. (4M)
- The ovule or megasporangium develops as a protuberance of placental tissue.
- A hypodermal cell (Nucellus) differentiate as archesporial cell.
- This cuts off some parietal cells and itself becomes megaspore mother cell.
- Megaspore mother cell undergoes meiosis forming a linear row of four megaspore of which three disintegrate and lowermost remains megaspore.
- Nucleus divides three times forming eight cells of which four move to each pole of embryo sac.
- One cell from each pole converges to centre to form secondary nucleus.
- Three cells towards micropyle form egg apparatus (1 egg and 2 synergids) and three cells on the opposite end become antipodal cells.
- Outermost cells from the base of ovule develop into integuments
Types of ovules
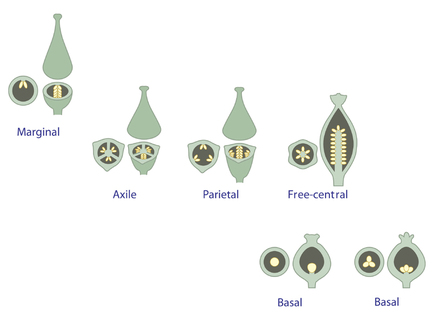
PLACENTATION
|
Figure: Ovary Placentation. Ovaries in cross section above ovaries in longitudinal section. A: marginal; B: parietal; C: axile, ovary with 2 loculi; D, axile, ovary with 3 loculi; E: free-central; F:free-central; G: apical; H:basal.
|
POLLINATION
- Definition: Pollination is the transfer of pollen grains from the anther of a flower to the stigma of the same flower or of another flower of same species.
- Types: The basic modes of pollination are
- A) Self pollination
- B) Cross pollination
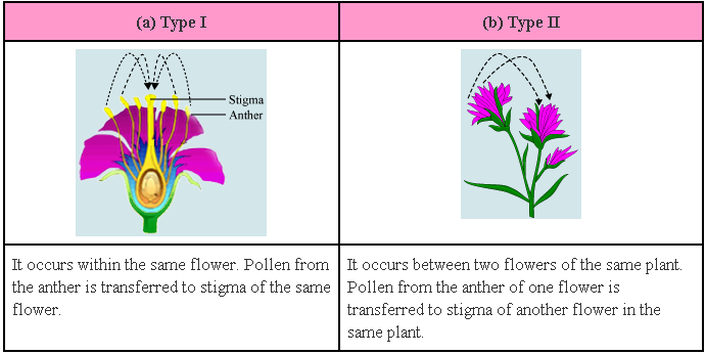
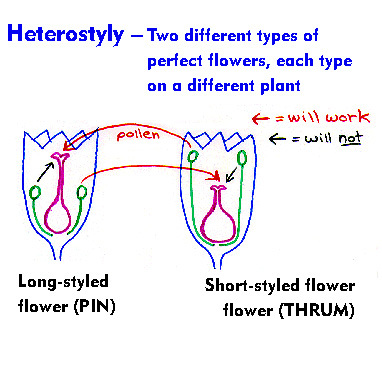
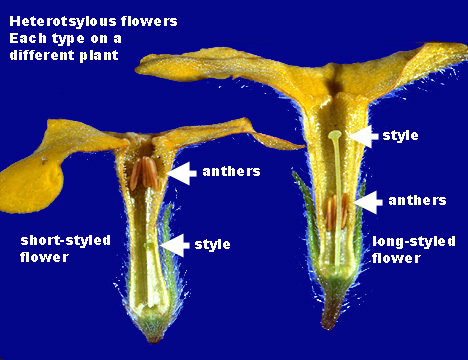
A) SELF POLLINATION :
- Transfer of pollen grains from the anther to stigma of the same flower or another flower on the same plant is said to be self pollination.
- Self pollination may autogamous or geitonogamous.
|
Commelina benghalensis : Both types of flowers
|
B) CROSS POLLINATION (xenogamy/allogamy)
- Transfer of pollen grains from the anther of a flower from one plant to the stigma of the flower on another plant is called cross pollination.
- It may need an external agency like wind or insects.
- Is geitonogamy a cross pollination?
Although geitonogamy is functionally cross-pollination involving a pollinating agent, genetically it is similar to autogamy since the pollen grains come from the same plant.
Contrivances of cross pollination
|
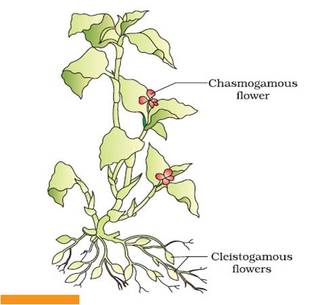
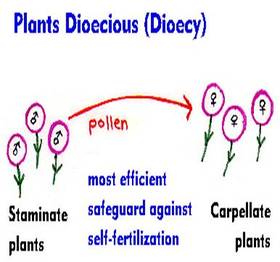
1. Unisexuality
|
|
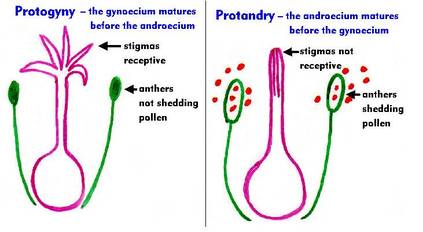
2. Dichogamy
|
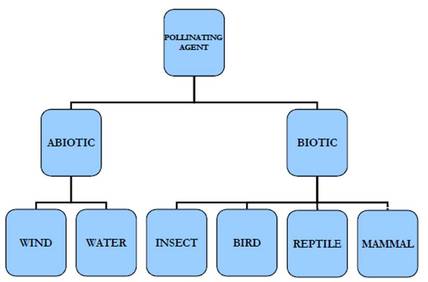
AGENTS OF POLLINATION
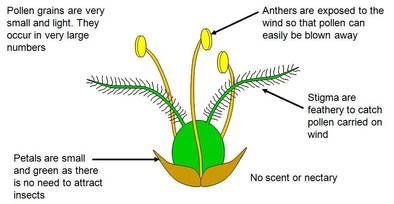
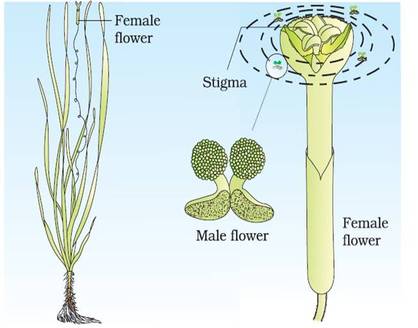
1. Pollination by wind (Anemophily)
|
2. Pollination by water (Hydrophily)
|
3. Pollination by insects
|
Special mechanism of insect pollination :
a) Salvia -
a) Salvia -
- It shows lever mechanism.
- Anthers are distractile ; lower lobe is sterile and upper lobe is fertile.
- The flower is bilabiate and protandrous.
- The insect sits on the lower lobe of the corolla; the upper fertile lobe of anther touches the body of insect.
- It ruptures and pollen grains are shed on the back of the insect.
- When this insect enters the other flower, pollination is affected
4. Pollination by birds(ornithophily)-
Figure : Pollination by Hummingbird
|
|
5. Pollination by bats (Chiropterophily)
Figure : Pollination by bat
|
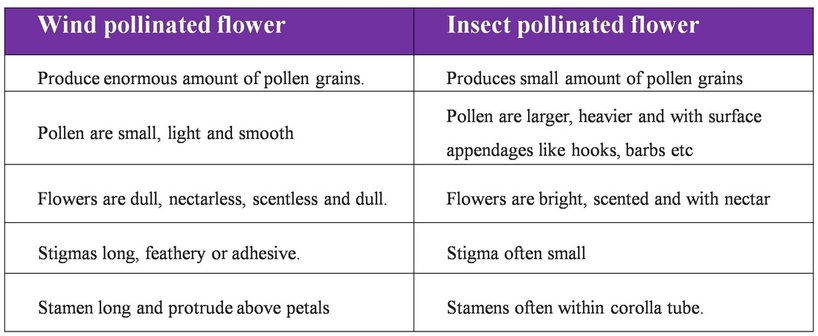
Difference between insect and wind pollinated flower
Difference between self and cross pollination
BQ: Write four advantages of cross pollination over self pollination. 2M (ISC 2009 )
- Cross pollination induces variations
- May eliminate harmful characters
- Result in hybrid formation
- More viable seed formed.
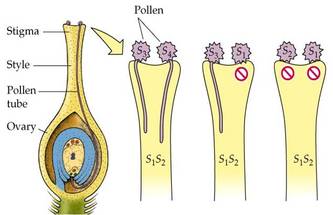
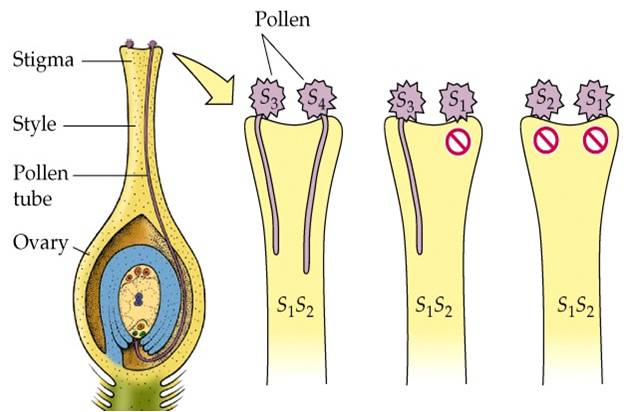
POLLEN PISTIL INTERACTION
|
2. Germination of
pollen and development of male gametophyte
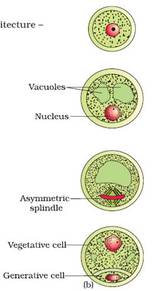
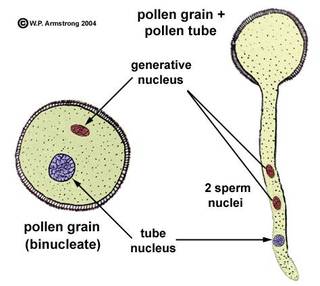
- The pollen grain germinates on the stigma to produce a pollen tube through one of the germ pores.
- The contents of the pollen grain move into the pollen tube
- In some plants, pollen grains are shed at two-celled condition (a vegetative cell and a generate cell). In such plants, the generative cell divides by mitosis and forms the two male gametes during the growth of pollen tube in the stigma.
- When the pollen is shed from anther it has usually two cells, a generative cells and a tube cell (Vegetative cell).
- In plants which shed pollen in the three-celled condition, pollen tubes carry the two male gametes from the beginning.
- Pollen tube, after reaching the ovary, enters the ovule.
3. Entry of pollen tube in ovule
- The pollen tube is directed towards one of the ovules.It may enter the ovules through one of the following three routes.
- Porogamy: When pollen tube enters the ovule through the micropyle,the condition is known as porogamy. This is the most common mode of pollen tube entry into the ovule.
- Chalaogmay :When pollen tube enters the ovule through the chalazal end, the condition is said to be chalazogamy. Eg. Betula, Casurina
- Mesogamy: When pollen tube enters the ovule through integument, the condition is known as mesogamy. Eg. Cucurbita and Populus
4. Entry of pollen tube in embryo sac
- Irrespective of the route of pollen tube into the ovule, it always enters the embryo sac from micropylar end.
- Recent studies have shown that filiform apparatus present at the micropylar part of the synergids guides the entry of pollen tube.
- The entry of pollen tube into the embryo sac may be
- Between the egg cell and one of the synergids
- Between the wall of the embryo sac and synergid.
- Between the synergids
- Directly penetrates one of the synergids
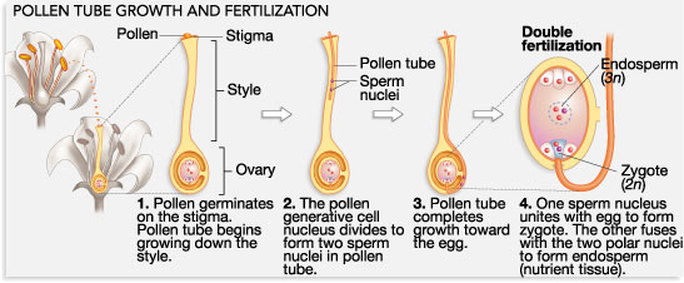
DOUBLE FERTILIZATION
|
|
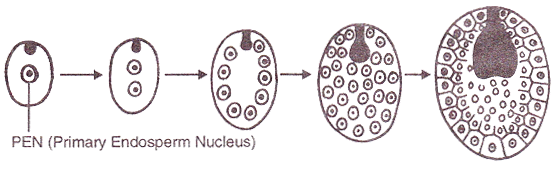
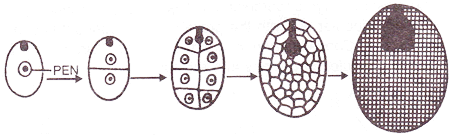
DEVELOPMENT OF ENDOSPERM
- The primary endosperm nucleus (3n) undergoes a series of divisions and ultimately forms endosperm, a highly nutritive tissue which provides nourishment to the developing embryo.
- Types of endosperm: On the basis of development, three types of endosperm have been recognised.
|
1. Nuclear endosperm:
|
|
3. Helobial endosperm
|
BQ: Give reason why endosperm in angiosperms becomes triploid. (ISC 2000: 3M)
BQ: Explain the development of the different types of endosperms in angiosperms. (ISC 2007,4M)
BQ: Explain the development of the different types of endosperms in angiosperms. (ISC 2007,4M)
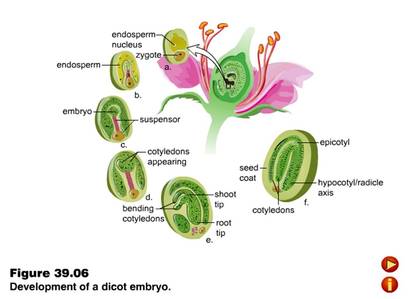
DEVELOPMENT OF EMBRYO
- After fertilization, a series of changes takes place in the ovule and as a result seed is is formed.
- The process of development of mature embryo from diploid zygote is called embryogenesis.
- Most zygotes divide only after certain amount of endosperm is formed. This is an adaptation to provide assured nutrition to the developing embryo.
- Though the seeds differ greatly, the early stages of embryo development (embryogeny) are similar in both monocotyledons and dicotyledons.
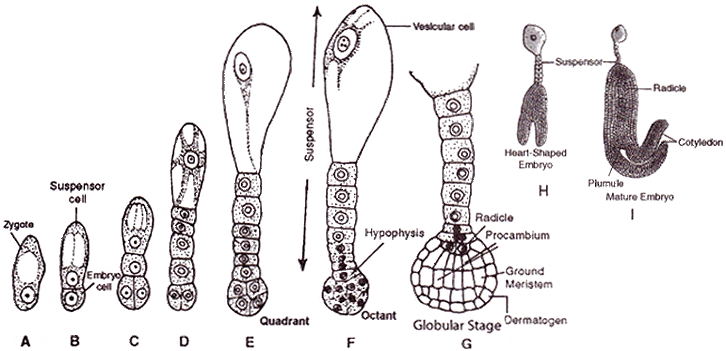
Development of dicotyledonous embryo :
- The normal type of dicot embryo development has been studied in Shepherd's purse (Capsella bursa-pastoris) which belongs to family Cruciferae. This is called as Crucifer or Onagrad type of embryo development.
- Zygote (oospore) divides into two unequal cells, larger suspensor cell towards micropyle and a smaller embryonal cell (terminal cell) towards antipodal region.
- The suspensor cell undergoes transverse divisions forming 6 - 10 celled long suspensor. The first cell of the suspensor (towards micropyle) is large and called haustorium or vesicular cell. The last cell of suspensor (towards embryo cell) is known as hypophysis. It forms radicle tip.
- Embryonal cell divides twice vertically and once transversely to produce a two-tiered eight-celled embryo. The epibasal tier forms two cotyledons and a plumule while the hypobasal tier produces only hypocotyl and most of the radicle.
- For this the octant embryo undergoes periclinal divisions producing protoderm, procambium and ground meristem.
- It is initially globular but with the growth of cotyledons it becomes heart-shaped and then assumes the typical shape, e.g., Capsella bursa-pastoris.
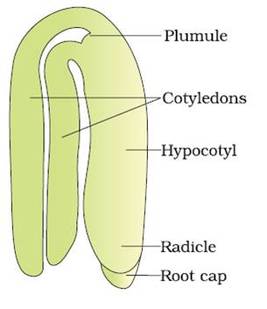
- A typical dicotyledonous embryo consists of an embryonal axis and two cotyledons.
- The portion of embryonal axis above the level of cotyledons is the epicotyl, which terminates with the plumule or stem tip.
- The cylindrical portion below the level of cotyledons is hypocotyl that terminates at its lower end in the radical or root tip.
- The root tip is covered with a root cap. (Figure: A typical dicot embryo)
BQ: Describe formation of embryo from a fertilized egg in angiosperm. 5M .(ISC 2003)
- Fertilized egg surrounds itself with a wall and becomes oospore.
- Oospore divides into suspensor cell and embryonal cell.
- Suspensor cell divides to form a row of cells which form the suspensor.
- Terminal cell of the suspensor near the micropyle enlarges and is known as haustorium.
- Basal cell of the suspensor is known as hypophysis cell.
- Embryonal cell divides and form eight celled octant.
- Four cells lying towards the suspensor is known as posterior octant which gives rise to radicle and hypocotyl.
- Anterior octant gives rise to plumule and cotyledons
- Hypophysis form the apex of radicle.
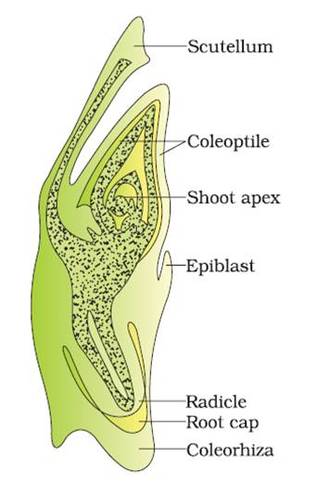
Development of Embryo in Monocot
|